Mean values of pH in CaCl2, saturation per base and potential acidity... | Download Scientific Diagram

Question Video: Calculating the Mass of Calcium Chloride That Contains a Given Mass of Chlorine | Nagwa

SOLVED: Choose the best classification of the reaction represented by the following equation: Pb(NO3)2(aq) + CaCl2(aq) → PbCl2(s) + Ca(NO3)2(aq) Multiple Choice decomposition acid-base oxidation-reduction neutralization precipitation

Mean values of pH in CaCl2, saturation per base and potential acidity... | Download Scientific Diagram

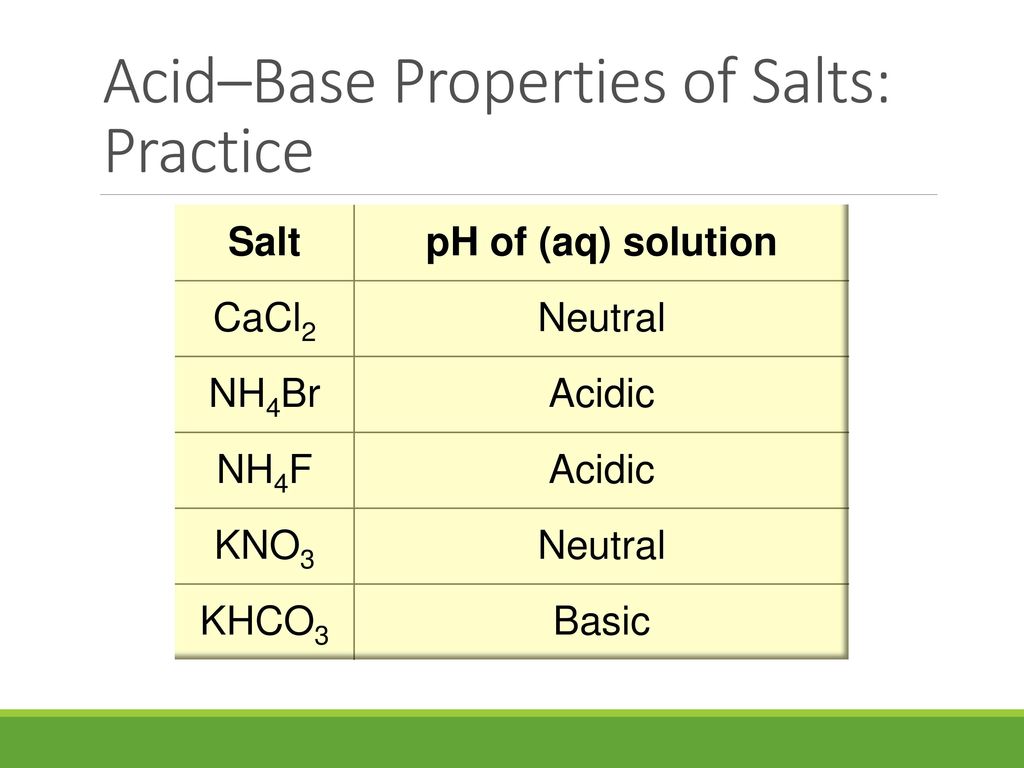
Name the acids and bases from which the following salts may be obtained. (i) Potassium sulphate (ii) Calcium chloride








![HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws] HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/equipment/pictures/calcium.hcl.gen.fig.gif)