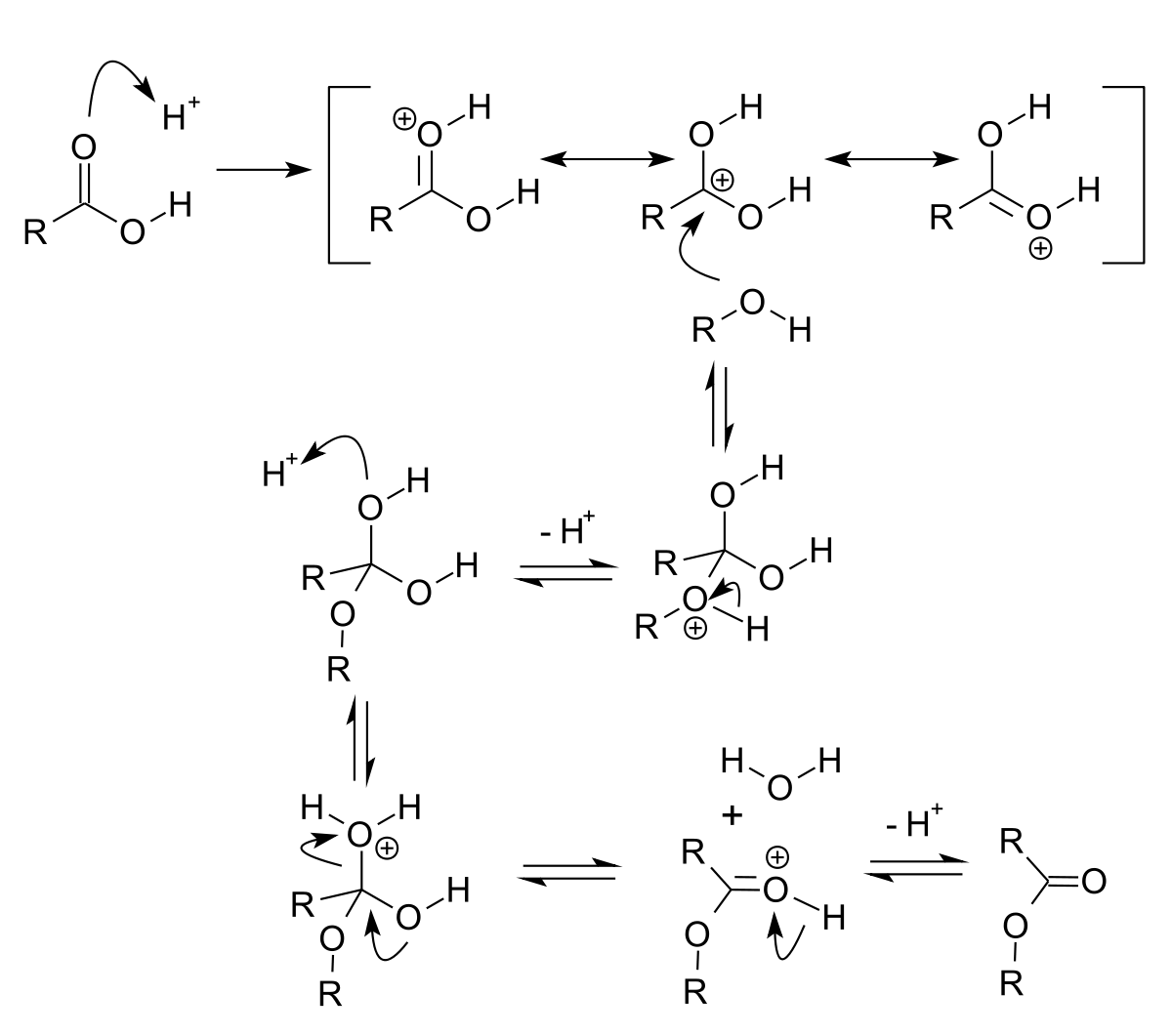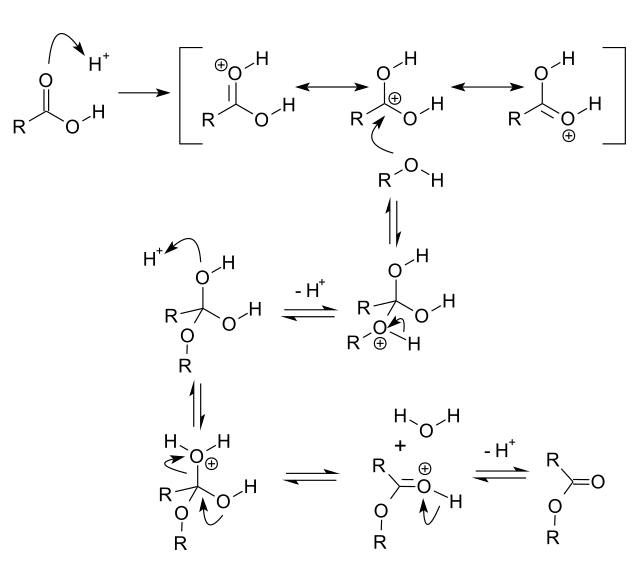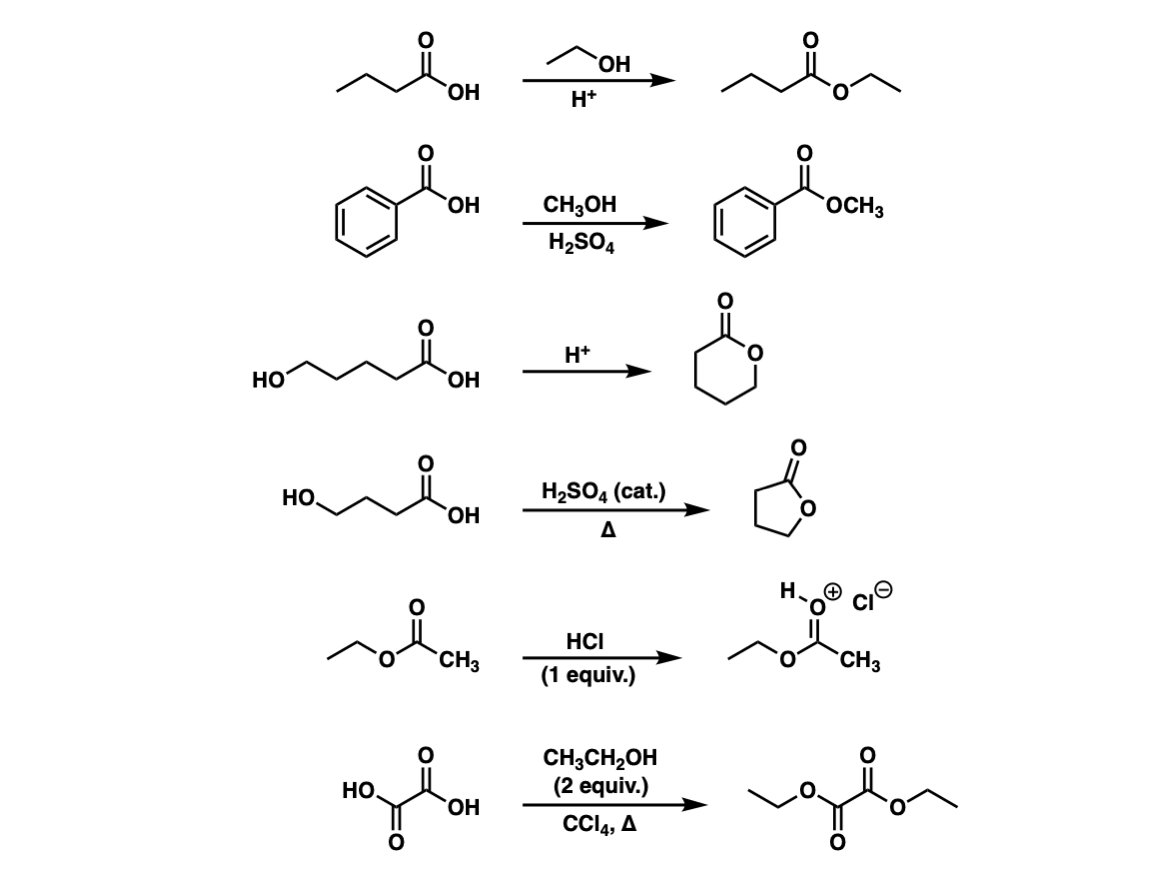
Conversion of carboxylic acids to esters using acid and alcohols (Fischer Esterification) – Master Organic Chemistry
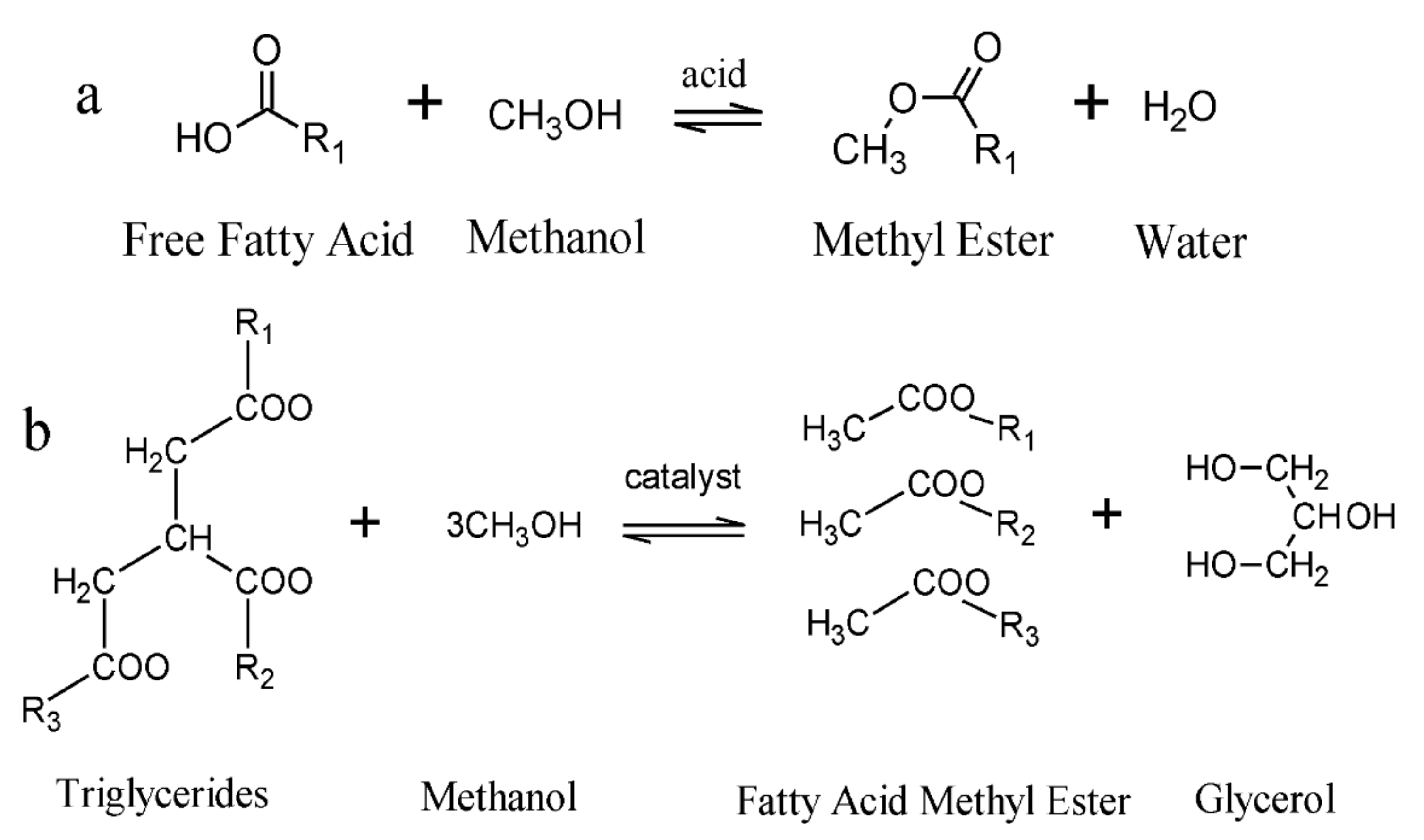
Catalysts | Free Full-Text | A Short Review on Catalyst, Feedstock, Modernised Process, Current State and Challenges on Biodiesel Production | HTML

Base-catalyzed selective esterification of alcohols with unactivated esters - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB02411A

How can you convert a carboxylic acid into an ester? a. Heat with an alcohol and catalytic acid. b. Deprotonate with a base and react with an alcohol. c. Deprotonate with a
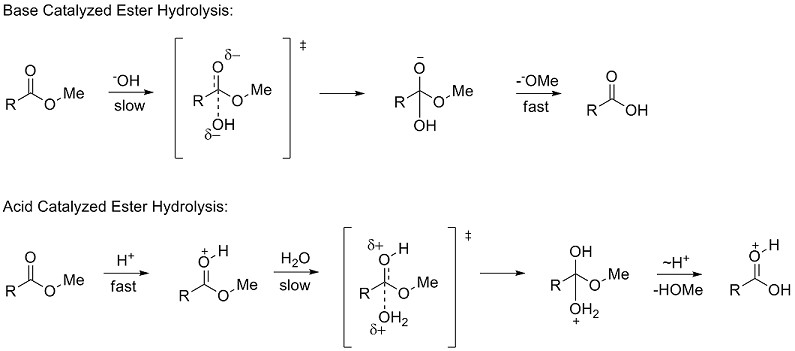
Structural Biochemistry/Organic Chemistry/Mechanisms of Organic Molecules - Wikibooks, open books for an open world

Why are the free fatty acids more easily converted into esters with a acid catalyzed method but not with the base catalyzed method? | Homework.Study.com