
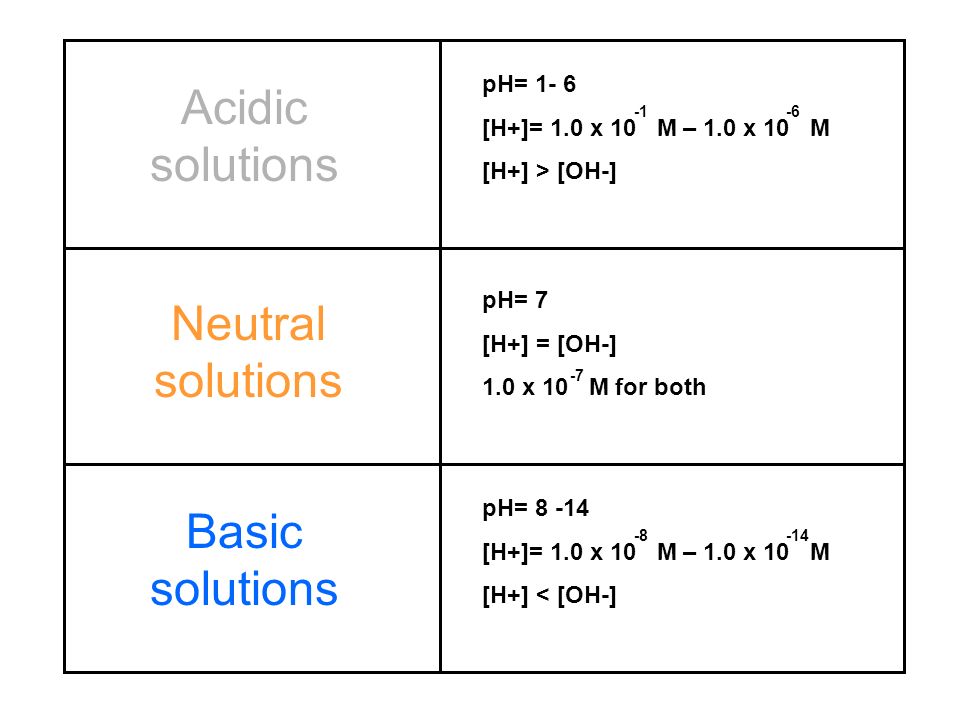
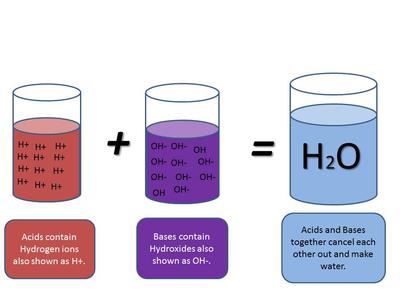
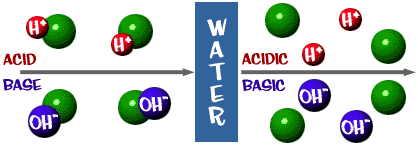
SOLVED: Acids ionize in water to produce H+. This H+ causes indicators like phenolphthaleinand litmus to change color. The un-ionized acid molecules have no effect on indicators.Ionic equations – total and net -
Identifying Acids and Bases Acids Acid (anhydrides) Bases Base (anhydrides) Salts contains H+ ions as the cation, with and other element as the anion. - ppt download

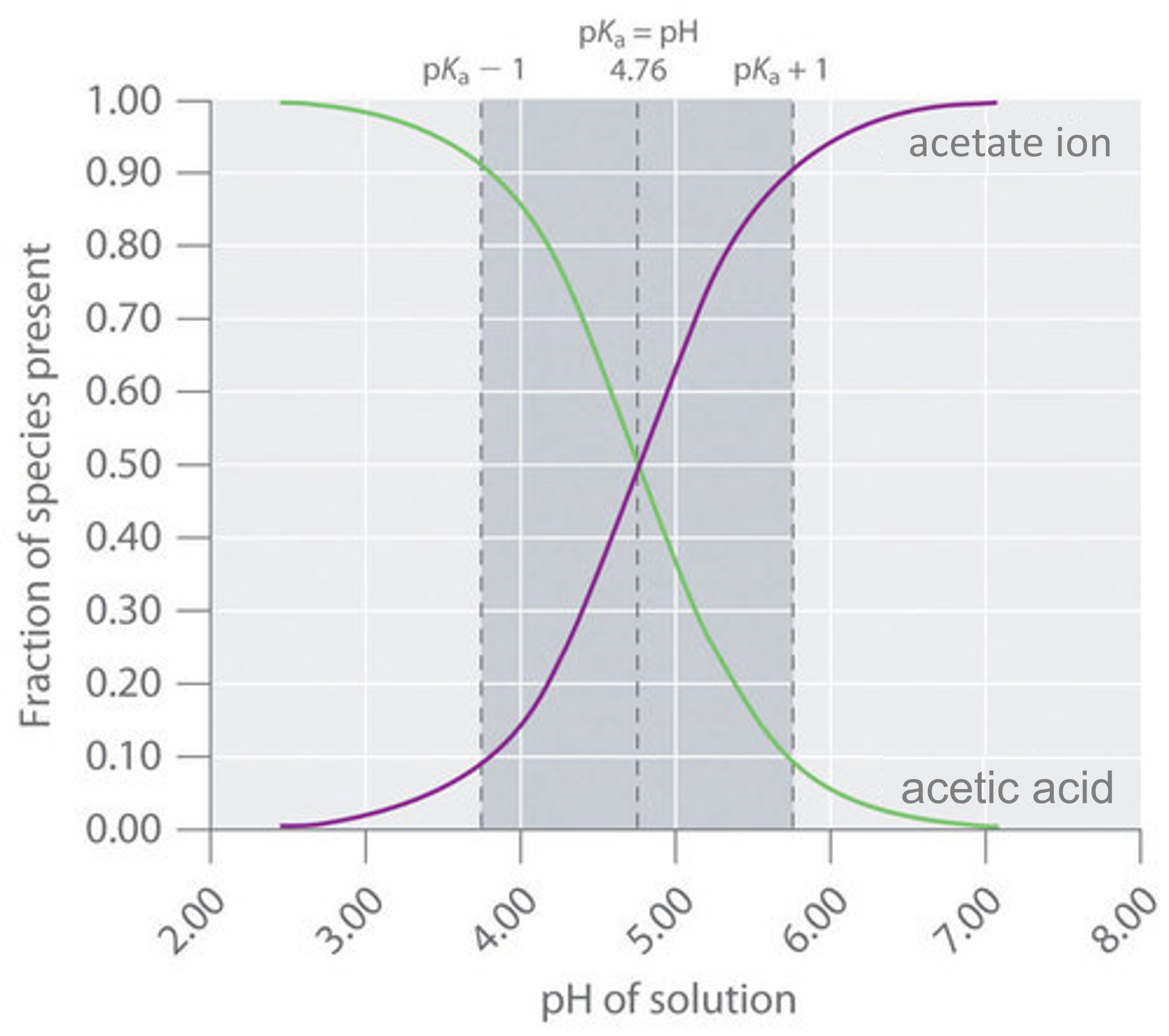
Determining PH of a Solution | Acidic, Basic & Neutral Solutions - Video & Lesson Transcript | Study.com









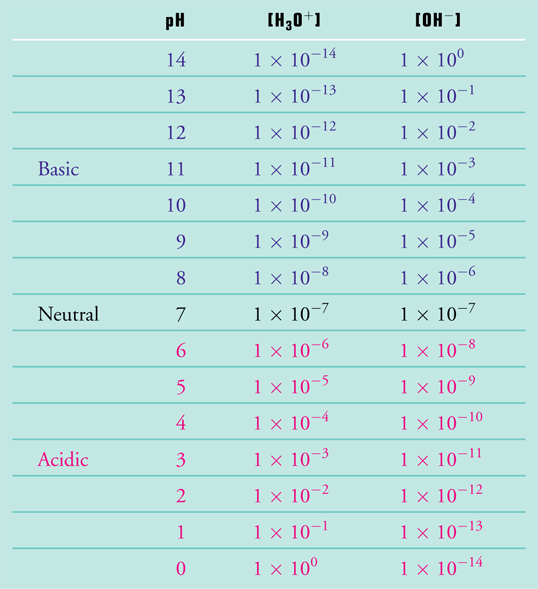
![Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube](https://i.ytimg.com/vi/UiK37I159fc/maxresdefault.jpg)




