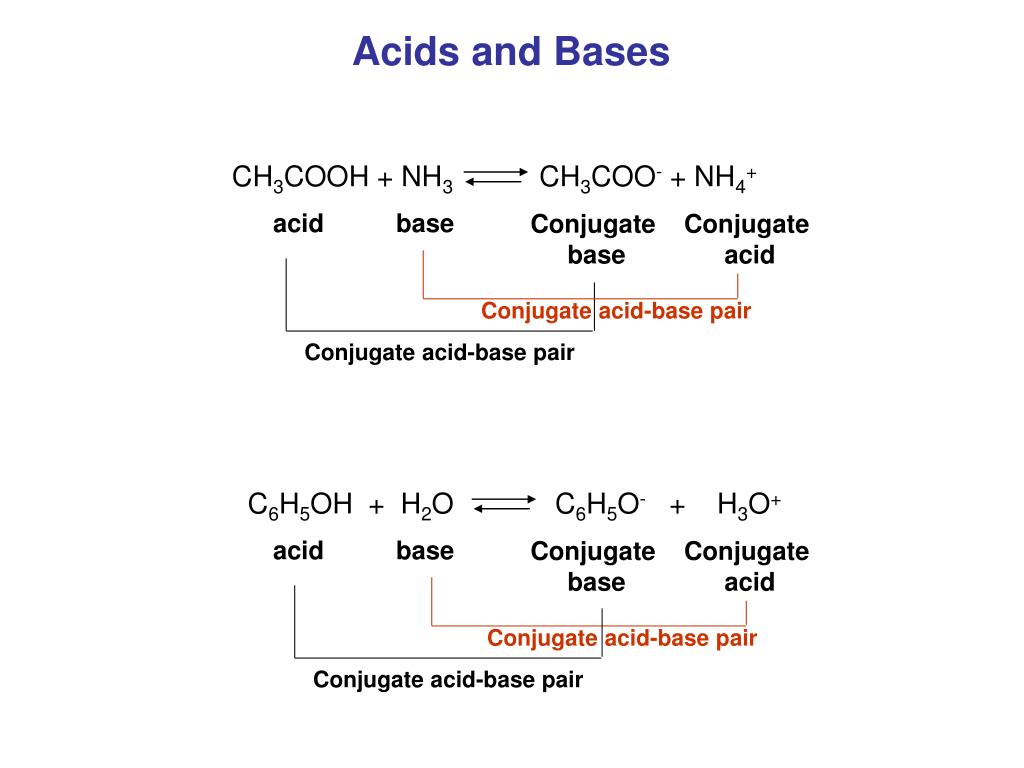
Why is oh- acid and h3o+ base H20(/) + H20(/) Acid Base H30+(aq) + OH-caq) Conjugate Conjugate base acid - Chemistry - Equilibrium - 13361499 | Meritnation.com

pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube


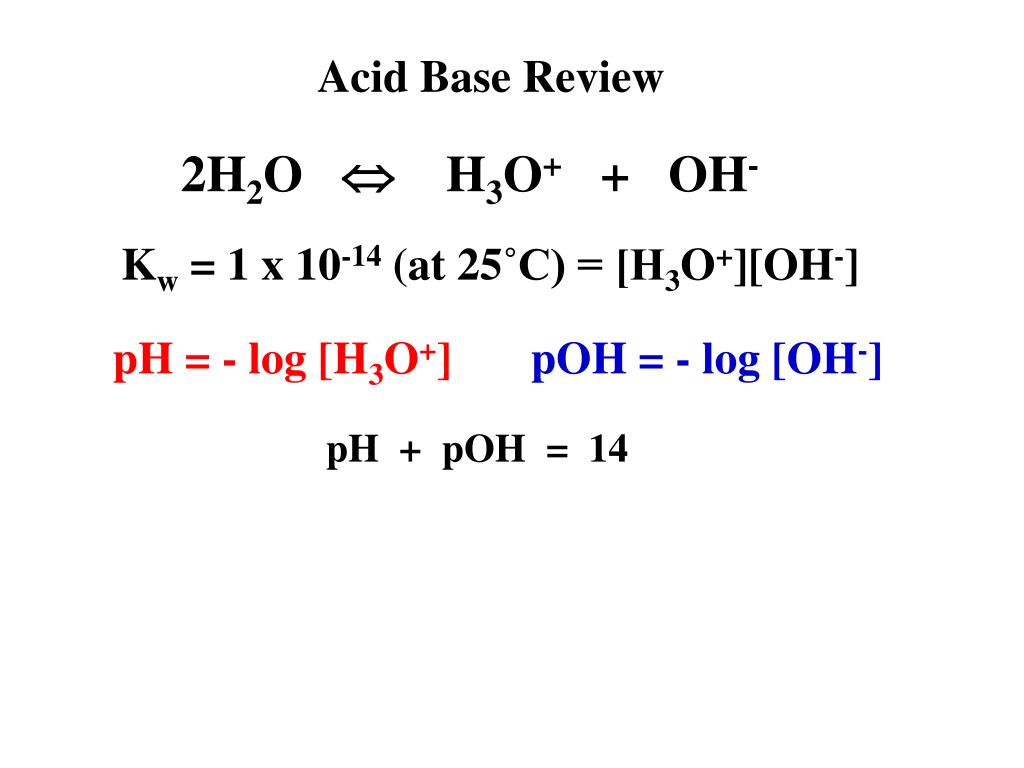
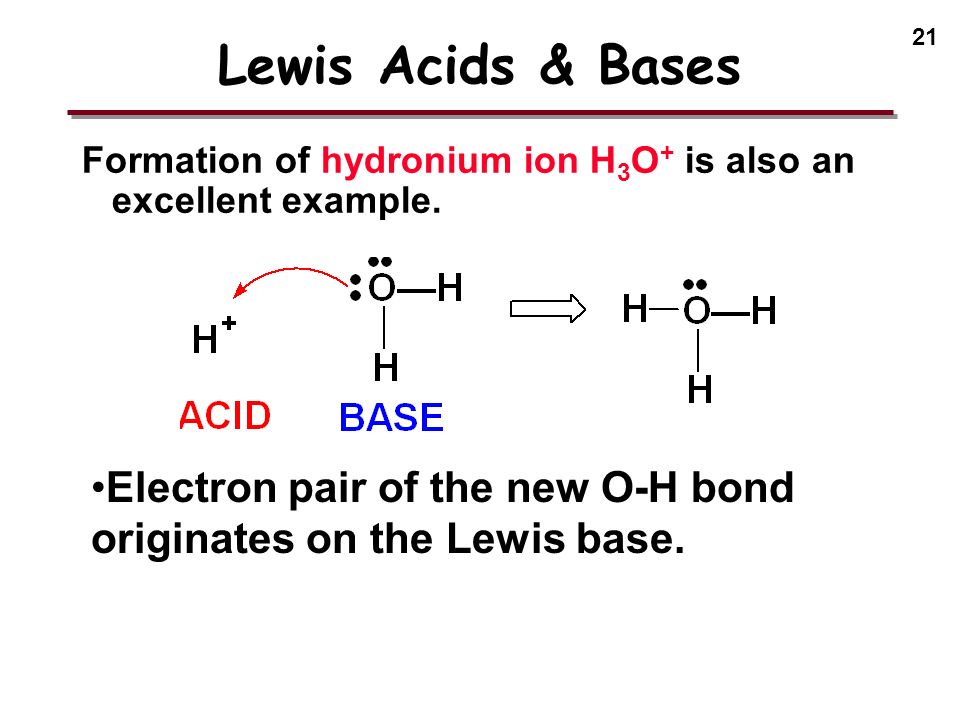
/chapter3/pages33and34/page33and34_files/aqh3o.png)
![Solved [H3O+] [OH-] pH pОН Acidic, Base or Neutral? | Chegg.com Solved [H3O+] [OH-] pH pОН Acidic, Base or Neutral? | Chegg.com](https://media.cheggcdn.com/media/f34/f34e841e-96b5-4eae-8369-3ddf8b4861c0/phpT8rw9L)
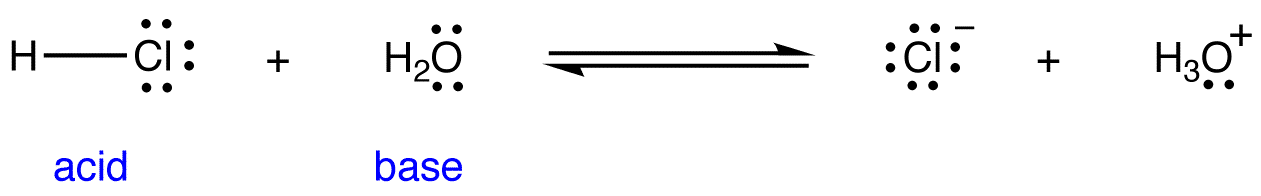

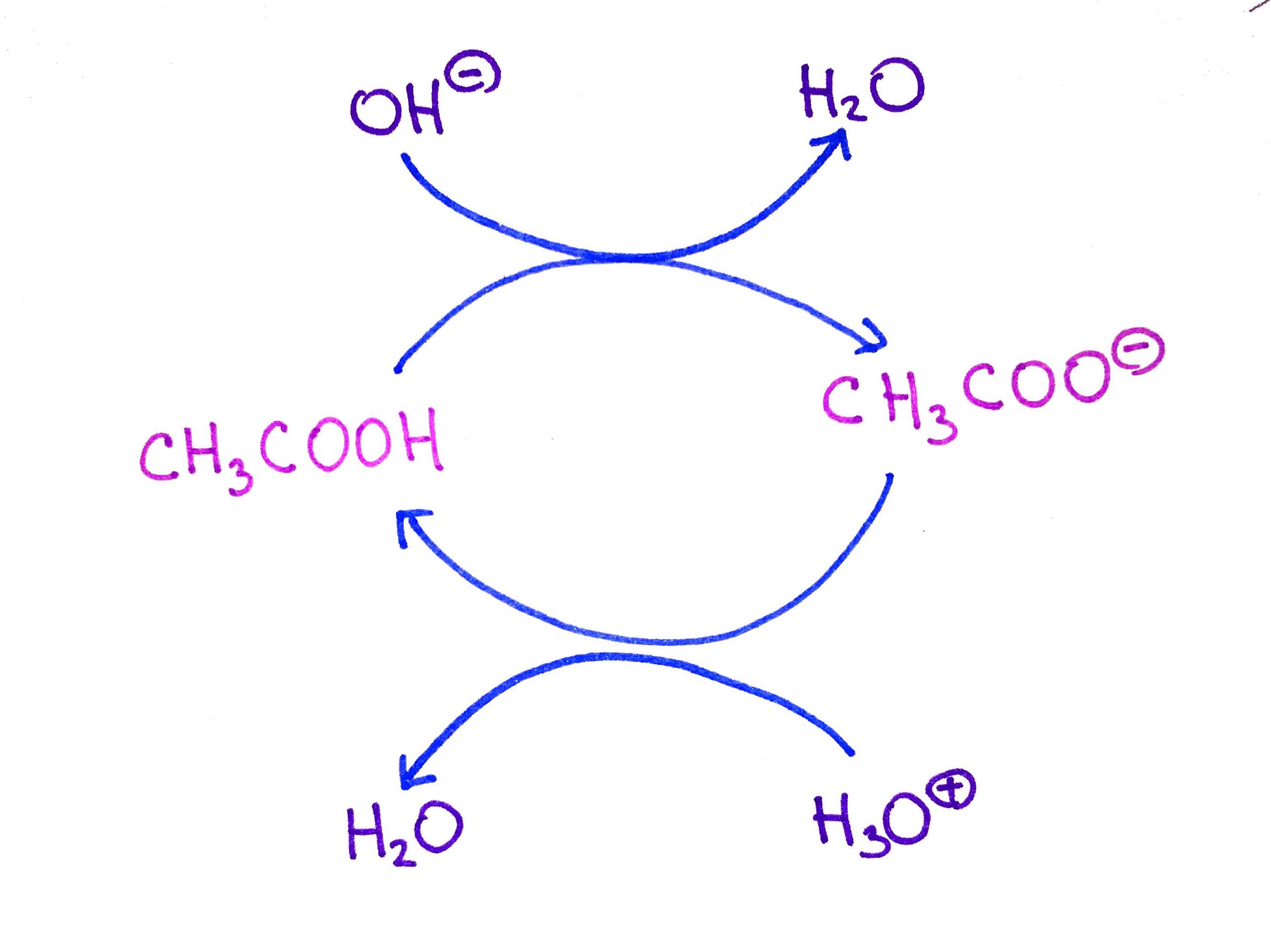
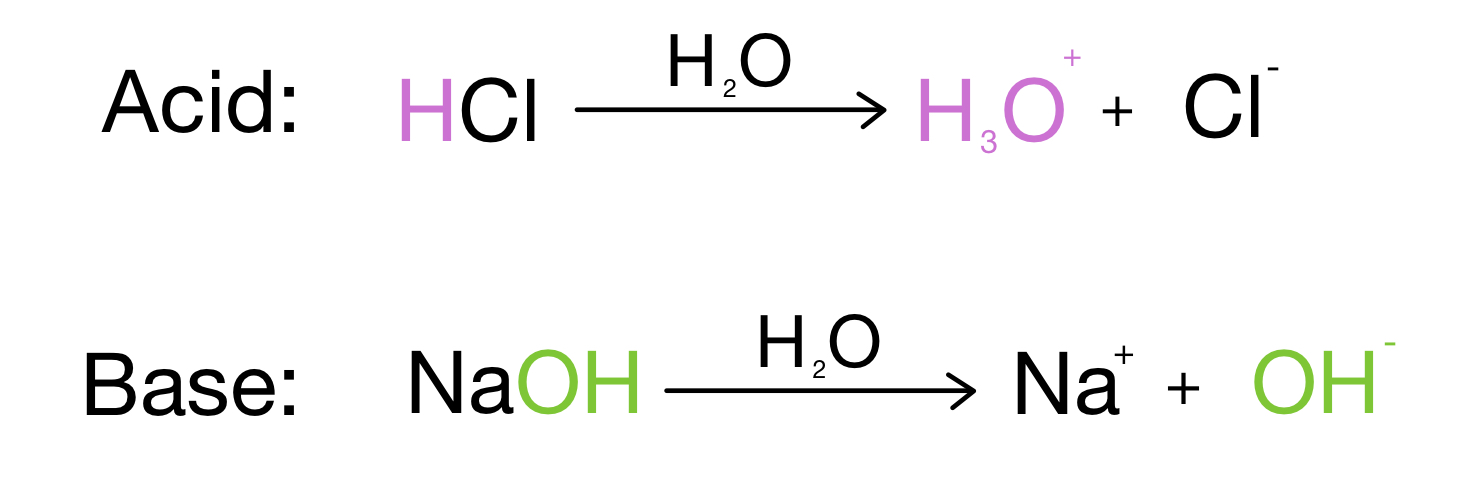
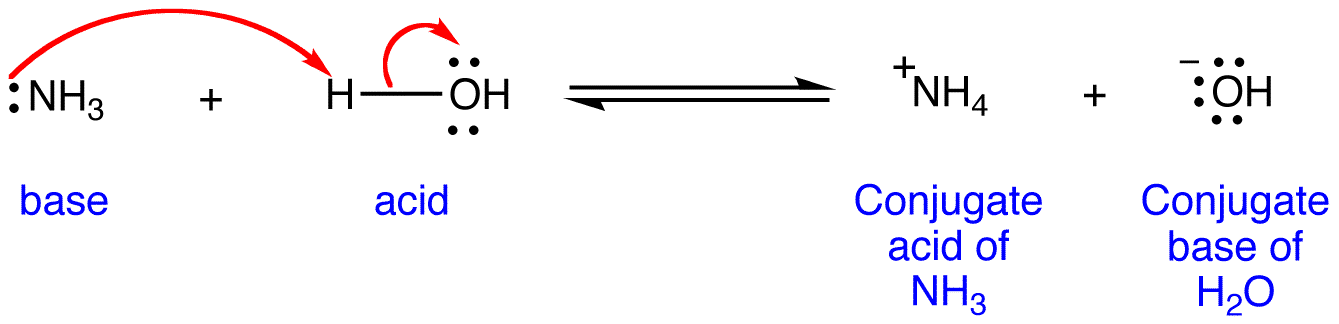
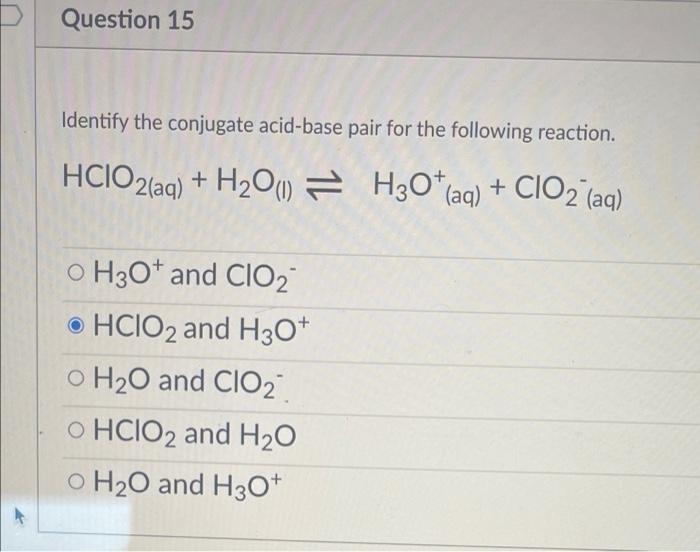
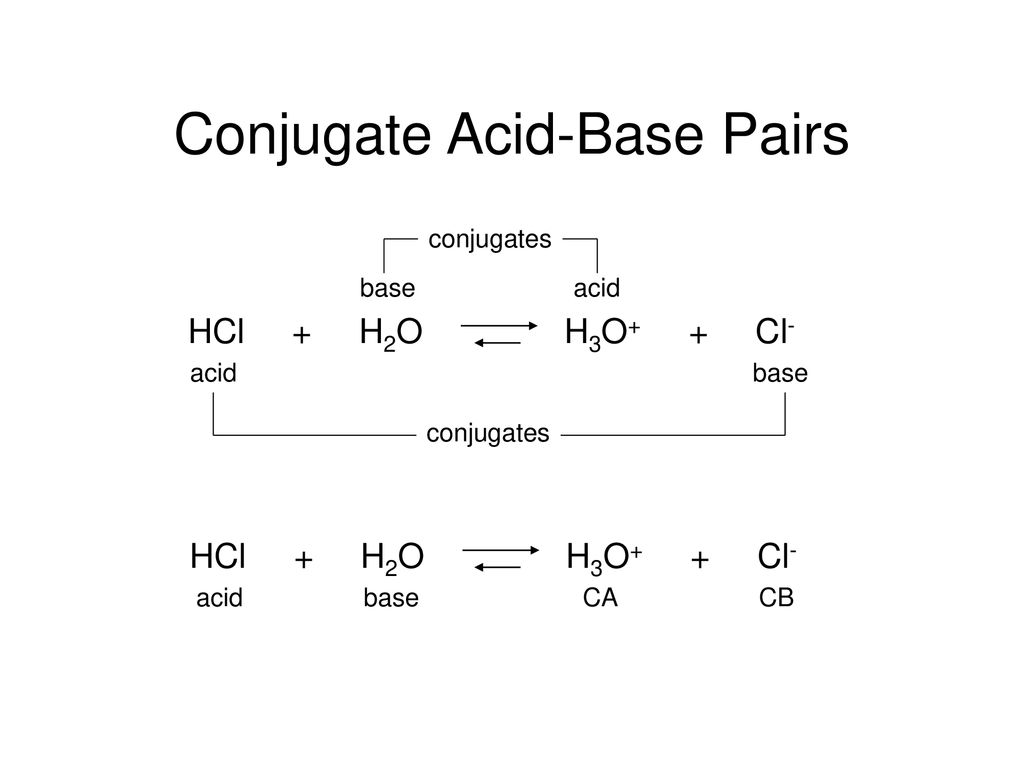
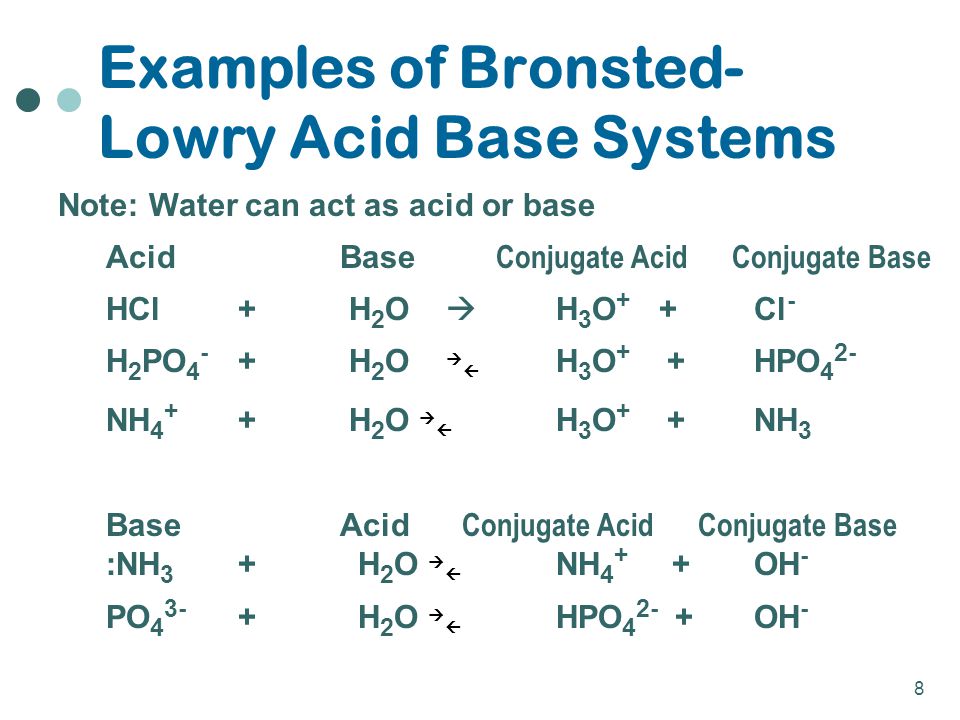

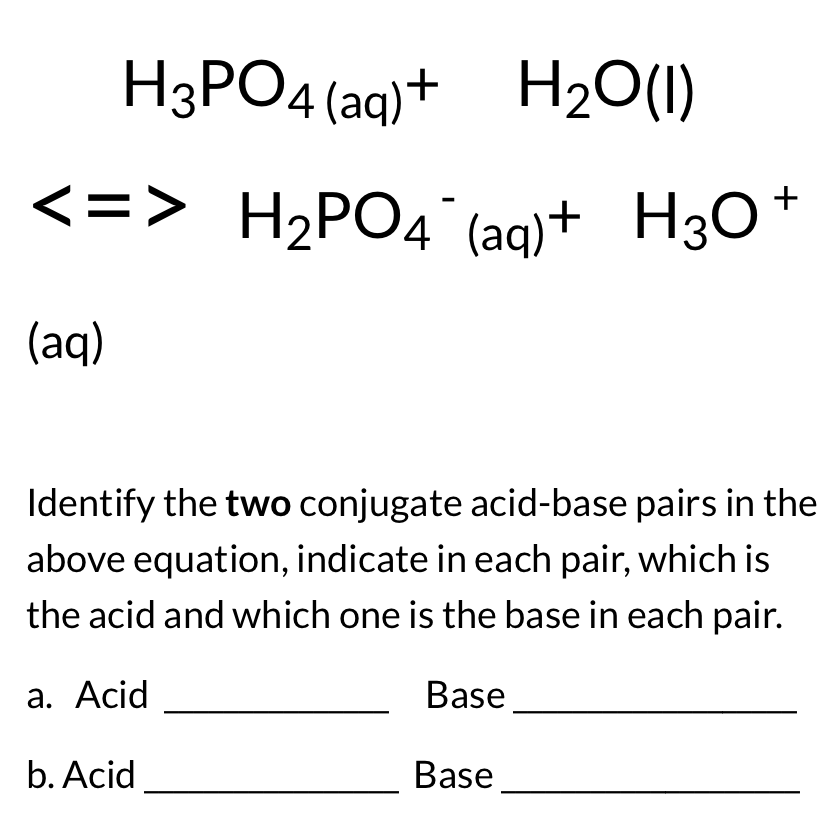
/chapter3/pages33and34/page33and34_files/ptsoh.png)

![Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube](https://i.ytimg.com/vi/UiK37I159fc/maxresdefault.jpg)